Saposin nanoparticles
Saposin nanodiscs
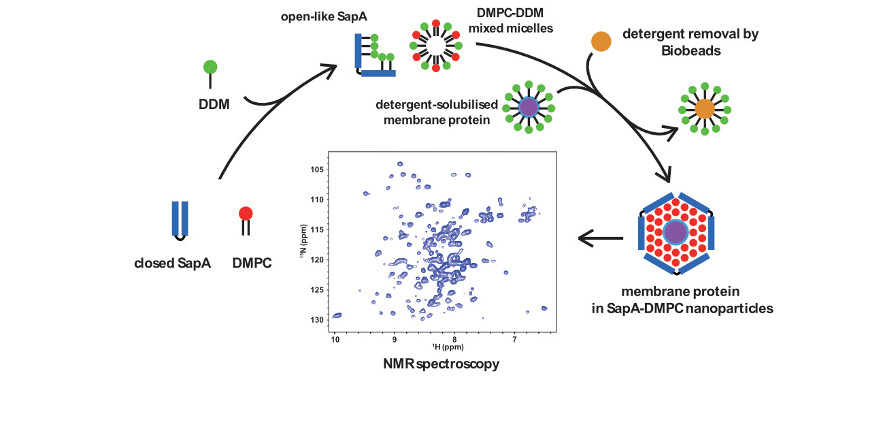 For membrane protein studies careful choice of membrane mimic is important. Detergents are the most commonly used medium, which are particularly suitable for NMR studies due to the relatively small size of detergent micelles as well as the range available and substantial research into their properties, which has led to a number of highly optimised detergents for membrane proteins. However, detergents do not mimic well many of the properties of lipid bilayers, such as the lateral pressure profile, and also have a number of disadvantageous properties for example they may disrupt protein-protein interactions, interfere with protein dynamics and denature soluble binding partners, or cytoplasmic regions of a protein.
For membrane protein studies careful choice of membrane mimic is important. Detergents are the most commonly used medium, which are particularly suitable for NMR studies due to the relatively small size of detergent micelles as well as the range available and substantial research into their properties, which has led to a number of highly optimised detergents for membrane proteins. However, detergents do not mimic well many of the properties of lipid bilayers, such as the lateral pressure profile, and also have a number of disadvantageous properties for example they may disrupt protein-protein interactions, interfere with protein dynamics and denature soluble binding partners, or cytoplasmic regions of a protein.
Consequently, other membrane mimics are sought of which lipid nanodiscs, have proved particularly popular. These consist of a membrane scaffold protein (MSP) which surrounds a bilayer of lipids where the membrane protein of interest is embedded; a variety of MSPs have been engineered to produce disks of different sizes suitable for different techniques and membrane proteins. We are investigating a new lipid nanoparticle system which instead of the MSP protein, uses SapA to surround the lipid bilayer. Compared to MSP nanodiscs the SapA system is size-adaptable enabling incorporation of different-sized membrane proteins using the same SapA scaffold protein. Previous studies have identified the utility of this system for cryo-EM and we have shown successful incorporation of sensory rhodopsin II, OmpX and the β1AR into this system for NMR studies.
For more information, see:
An adaptable phospholipid membrane mimetic system for solution NMR studies of membrane proteins.
Chih-Ta Henry Chien, Lukas R. Helfinger, Mark J. Bostock, Andras Solt, Yi Lei Tan, and Daniel Nietlispach, Journal of the American Chemical Society(2017) 139, 14829-14832
(doi: 10.1021/jacs.7b06730)
